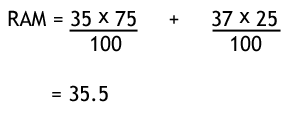The Atom
Edited by Jamie (ScienceAid Editor), Taylor (ScienceAid Editor)
About The Atom
An atom is the smallest unit of a particular substance. Below is a diagram of what an atom looks like:
The red and green circles in the centre are the proton and neutron, they make up the nucleus. The blue circle is the electron and the black ring shows its orbit around the electron takes around the nucleus. Now you need to know about the relative mass and charge of each of these components of the atom. This useful little table should help you to remember them:
| Particle | Charge | Mass |
|---|---|---|
| Proton | +1 | 1 |
| Electron | -1 | Negligible (0.00054) |
| Neutron | 0 | 1 |
Some Terms
There are some terms that you will need to know, and the meaning of them.
- 1Also called proton number, this is the number of protons the atom has. This can be looked up on the periodic table of the elements.Atomic number.Advertisement
- 2This basically tells you the number of particles in the nucleus (as the electron has negligible mass).Mass number.
These numbers, along with the element symbol are often written together as follows.
The top number is the mass number and the bottom is the atomic/proton number. If you want to work out the number of neutrons you simply subtract the bottom from the top, leaving you with 30.
Isotopes
An isotope is merely where you have different chemicals of the same element, but have different numbers of neutrons. i.e.. the atomic numbers are the same, but the mass is not. On the periodic table, the relative atomic mass is given. This can be thought of as the average mass of the atoms. The example below shows how to calculate this given the relevant data, regarding chlorine.
| Isotope | Mass number | Abundance |
|---|---|---|
| Chlorine - 35 | 35 | 75% |
| Chlorine - 37 | 37 | 25% |
And the answer is ...
Referencing this Article
If you need to reference this article in your work, you can copy-paste the following depending on your required format:
APA (American Psychological Association)
The Atom. (2017). In ScienceAid. Retrieved Apr 24, 2024, from https://scienceaid.net/chemistry/fundamental/atom.html
MLA (Modern Language Association) "The Atom." ScienceAid, scienceaid.net/chemistry/fundamental/atom.html Accessed 24 Apr 2024.
Chicago / Turabian ScienceAid.net. "The Atom." Accessed Apr 24, 2024. https://scienceaid.net/chemistry/fundamental/atom.html.
If you have problems with any of the steps in this article, please ask a question for more help, or post in the comments section below.
Comments
Article Info
Categories : Fundamental
Recent edits by: Jamie (ScienceAid Editor)