Isomerism
Edited by Jamie (ScienceAid Editor), Taylor (ScienceAid Editor), Jen Moreau
An isomer is a different arrangement of a molecule. They are especially important in Organic Chemistry because there are so many of them.
Structural Isomerism
These are compounds with the same molecular formula but different structures. This means that they have the same atoms and bonds, but they are in different places.
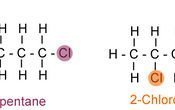
- 1The most common type of structural isomer is a chain isomer. This is where you have a methyl|methyl, etc., branching off of the main chain. There are actually slight differences in the physical properties of this type of isomer. The branched isomers, have weaker Van der Waals forces and therefore have a lower boiling point.Chain Isomers.Advertisement
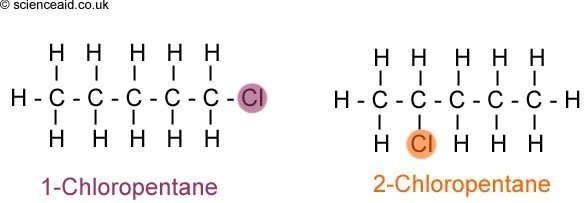
- 2The second type of isomer is a position isomer. This is where the functional group is in a different place. An easy to illustrate example is with haloalkanes. The functional group, in this case - Cl, is in a different place, therefore the above diagram shows two different position isomers.Position Isomers.
Structural and Geometrical Isomerism
Structural Isomerisms
A structural isomer is one of three types of isomerism where the bonding is changed. This includes:
- 1This is when branching occurs e.g. methylbutane.Chain Isomerism.
- 2These have the functional group in a different place (DO NOT confuse this with the next type). #Functional Group. These isomers have the same atoms, but they are bonded in such a way to create a different functional group. For example: Carbonyls.Positional Isomers.
Geometric Isomerisms
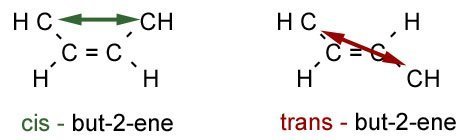
Geometric isomerism is a type of stereoisomerism, where there are different spatial arrangements of functional groups relative to the "double bond". There are two types, depending on whether the functional group is on the same of opposite side of the double bond.
- 1On the left is a cis isomer, where the two groups are on the same side of the double bond. This type of isomer usually has a slightly higher boiling point.Cis Isomer.
- 2On the right is a trans isomer, where the two groups are on opposite sides of the double bond. (think of transatlantic where you cross to the opposite side of the ocean). It will have a lower boiling point because it is less polar.Trans Isomer.
Stereoisomerism
There is also stereoisomerism, which is a different arrangement of molecules in 3D space. One type you have already encountered: geometrical isomerism, also known as cis-trans isomerism; resulting from the lack of rotation on a C=C bond. However, there is a further type of stereoisomerism known as "optical isomerism".
Optical Isomerism
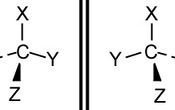
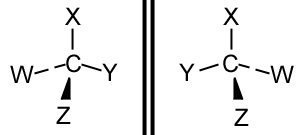
If a carbon atom has 4 different groups bonded to it, it is said to be chiral and has an asymmetric carbon. For example, see the molecule below.
Optical isomers tend to be mirror images of each other. You may think the molecules above are both the same, but if you construct them using a model set for example, you will see they can not be superimposed on each other. Optical isomers are known as enantiomers. Optical isomers are practically the same molecule (some are treated differently by enzymes).
Except when plane-polarized light is passed through a solution, one enantiomer will rotate the light in one direction, and the other in the opposite direction.Yet, if there are equal amounts of the two enantiomers, it creates a racemic mixture or a racemate. If plane-polarized light is beamed through this, there will be no effect since the solution has no optical activity. Racemates form in addition reactions on planar double bonds (like C=C or C=O), since attack might occur on either side of the plane.
Referencing this Article
If you need to reference this article in your work, you can copy-paste the following depending on your required format:
APA (American Psychological Association)
Isomerism. (2017). In ScienceAid. Retrieved Apr 23, 2024, from https://scienceaid.net/chemistry/organic/isomerism.html
MLA (Modern Language Association) "Isomerism." ScienceAid, scienceaid.net/chemistry/organic/isomerism.html Accessed 23 Apr 2024.
Chicago / Turabian ScienceAid.net. "Isomerism." Accessed Apr 23, 2024. https://scienceaid.net/chemistry/organic/isomerism.html.
If you have problems with any of the steps in this article, please ask a question for more help, or post in the comments section below.
Comments
Article Info
Categories : Organic
Recent edits by: Taylor (ScienceAid Editor), Jamie (ScienceAid Editor)