Nuclear Magnetic Resonance Structure Determination
Edited by Jamie (ScienceAid Editor), Taylor (ScienceAid Editor)
About NMR Spectroscopy
Hydrogen atoms have the property of spin, which means they have a magnetic field. NMR spectroscopy is used to give information about the relative numbers and positions of hydrogen atoms because the magnetic field of the hydrogen can either be aligned or not with an external field.
An NMR spectrum is obtained by dissolving a sample in a solvent that is deuterated meaning it has no protons that would interfere with the result, for example CCl4. This sample is then placed in a giant magnet and the test is run.
Structure Determination
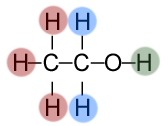
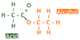
Different peaks are produced on the spectrum because resonances are produced depending on the particular chemical environments that the protons are in. To understand this, have a look at the ethanol molecule below.
Ethanol has three different chemical environments. The first is highlighted in red, as all three hydrogen atoms are bonded to a carbon that is bonded to CH2OH. And so the blue and green hydrogens are also separate chemical environments. Each of these three will resonate at different frequencies and produce separate peaks.
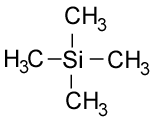
An NMR spectrum uses the molecule tetramethylsilane (TMS, below) as a standard, which means that all resonances are referenced from this point. This gives the chemical shift: δ which is the movement caused by shielding. It is measured in parts per million (ppm).
There are several reasons why this molecule is used. Firstly, it gives a resonance higher than almost all organic compounds and this peak is intense because it has 12 hydrogens with the same chemical environment. TMS is non-toxic and inert, so is safe to work with, and it has a low boiling point (26.5°C) so can be easily removed by distillation.
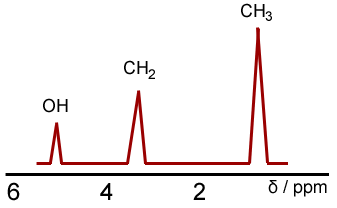
A useful feature of NMR spectra is that they give an indication of the numbers of hydrogen atoms in each environment. This can be done by two ways. Firstly, the integration values. The area under each peak will indicate how many atoms there are, relative to each other. So taking our example of ethanol, the following will be observed. Where the area under the CH3 peak is three times that of the OH peak.
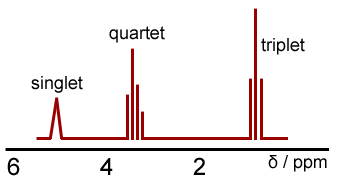
Similar information can be obtained using high resolution NMR spectra. Here, we see individual peaks being split in to several peaks, this is as a result of spin-spin couplings where the magnetic fields of neighbouring protons interact. Again, let's look at a high resolution NMR of ethanol.
The splitting of the peaks occurs according to the n+1 rule. Where a peak splits in to, one more than the number of protons in the group next to it, peaks. For example, take a look at the CH2 group, it is next to a CH3, which has 3 protons; therefore it splits to 4 (a quartet).
Interpreting NMR Spectra
You now know the three features of the NMR spectrum that will help you determine the structure of an organic compound.
- 1First, you can check the chemical shift of the peak, which will indicate the group.Chemical Shift.Advertisement
- 2The integration ratios will give an indication of how many protons there are in that particular group.Integration Ratios.
- 3The splitting patterns of the peaks will give an indication of where each group is, especially if you already know the integration ratios.Splitting Patterns.
Referencing this Article
If you need to reference this article in your work, you can copy-paste the following depending on your required format:
APA (American Psychological Association)
Nuclear Magnetic Resonance Structure Determination. (2017). In ScienceAid. Retrieved Apr 26, 2024, from https://scienceaid.net/chemistry/organic/nmr.html
MLA (Modern Language Association) "Nuclear Magnetic Resonance Structure Determination." ScienceAid, scienceaid.net/chemistry/organic/nmr.html Accessed 26 Apr 2024.
Chicago / Turabian ScienceAid.net. "Nuclear Magnetic Resonance Structure Determination." Accessed Apr 26, 2024. https://scienceaid.net/chemistry/organic/nmr.html.
If you have problems with any of the steps in this article, please ask a question for more help, or post in the comments section below.
Comments
Article Info
Categories : Organic
Recent edits by: Jamie (ScienceAid Editor)