Analysing Gases
Edited by Jamie (ScienceAid Editor), Taylor (ScienceAid Editor), SmartyPants
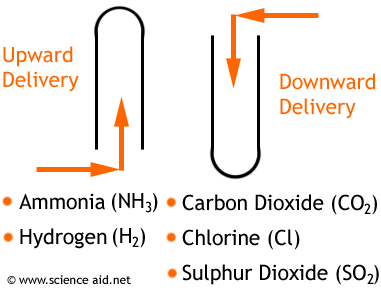
Methods of Collection
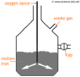
- 1One way for gasses to be collected is over water, but this can only be done with gasses that are insoluble or just slightly soluble in water, otherwise they would dissolve and you wouldn't be able to collect them. For example: carbon dioxide, oxygen, and hydrogen. For this method, a container is filled with water, a test tube is also partially filled with water and inverted and submerged in the water - it remains full. A pipe is put under the test tube, and as gas bubbles up into the test tube, it forces water from it; so that the gas is collected in the tube.Over Water.Advertisement
- 2
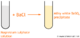
- 3The next method of collection is the gas syringe. It's not only for collecting gasses, but also for measuring the volume of the gas collected.Syringe.
Testing
| Gas | Testing Method |
|---|---|
| Hydrogen
H2 |
Collect gas
Put in a burning match. It will pop. |
| Oxygen
O2 |
Collect gas.
Put in a glowing match. It will reignite. |
| Carbon Dioxide
CO2 |
Bubble gas through lime water (CaOH).
The water will turn cloudy. |
| Sulphur Dioxide
SO2 |
Collect gas.
Bubble through orange potassium dichromate solution. Solution turns green. |
| Hydrogen Chloride
HCl |
Collect gas.
Mix with ammonia gas. Forms dense white fumes. |
| Ammonia
NH3 |
Collect gas
Add damp red litmus paper. Paper turns blue. |
Referencing this Article
If you need to reference this article in your work, you can copy-paste the following depending on your required format:
APA (American Psychological Association)
Analysing Gases. (2017). In ScienceAid. Retrieved Apr 27, 2024, from https://scienceaid.net/chemistry/applied/gases.html
MLA (Modern Language Association) "Analysing Gases." ScienceAid, scienceaid.net/chemistry/applied/gases.html Accessed 27 Apr 2024.
Chicago / Turabian ScienceAid.net. "Analysing Gases." Accessed Apr 27, 2024. https://scienceaid.net/chemistry/applied/gases.html.
If you have problems with any of the steps in this article, please ask a question for more help, or post in the comments section below.
Comments
Article Info
Categories : Applied
Recent edits by: Taylor (ScienceAid Editor), Jamie (ScienceAid Editor)