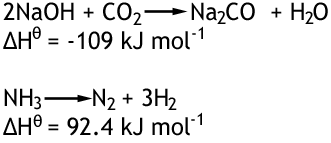Enthalpy Change
Edited by Jamie (ScienceAid Editor), Taylor (ScienceAid Editor)
What is Enthalpy?
Usually there is energy change involved in a chemical reaction, somehow. In chemistry, this change in heat energy is represented as enthalpy change using the following symbol:
So that this figure is the same everywhere and fair, it is taken at standard conditions which are 298K and 100 kPa pressure. Here are two reactions, each with a different enthalpy change.
- 1This is because the reaction is exothermic meaning it gives out heat. The reason it is negative is energy is leaving the reaction.For the first reaction, the enthalpy change is negative.Advertisement
- 2The second reaction has a positive enthalpy change, this is because it is endothermic and heat is entering the system.
Enthalpy of Reaction, Combustion, and Formation
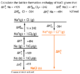
These are three different types of situations that an energy change may take place. On the top image x is used to show where a letter is written to indicate what type of enthalpy is shown: r for reaction and so on.
- 1The easiest one to get your head around is standard enthalpy of reaction. This is merely the energy change of a reaction in the quantities that are expressed.Reaction.
- 2The standard enthalpy of combustion is the enthalpy change when 1 mole of substance is completely burned in excess oxygen under standard conditions, all reactants in their standard states. The enthalpies of combustion can be determined using a calorimeter which is explained below; it uses the symbol c.Combustion.
- 3The standard enthalpy of formation is the enthalpy change when 1 mole of compound is produced from its elements in standard conditions, all products and reactants in the standard state. The standard state is important, it means the way that the element is, at standard conditions (see above). So you would have H2(g) NOT H on its own. Therefore you sometimes need to do fractions of elements, as with the standard enthalpy of formation of ethanol: C (s) + 2H2 (g) + ½O2 (g) ® CH3OH (l)Formation.
Calorimetry
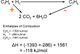
This is a method we can use to find out what the enthalpy changes of reactions are. Typically you will do this at school using some sort of alcohol heating up some water. To do it correctly, you would have it in a closed system so no heat escapes, but this is not usually possible in a high school lab. Once the water has been heated up, you must use the following equation.
A bit more about the specific heat capacity this is the energy required to raise the temperature of one gram of substance by 1 kelvin (or celsius depending on what you're using). It is measured in Jg-1K-1 and water has a specific heat capacity of 4.18 Jg-1K-1. For example, if 1g of ethanol is burned to heat 100g of water, raising its temperature by 42K, you would do the following calculations:
Referencing this Article
If you need to reference this article in your work, you can copy-paste the following depending on your required format:
APA (American Psychological Association)
Enthalpy Change. (2017). In ScienceAid. Retrieved Apr 28, 2024, from https://scienceaid.net/chemistry/physical/enthalpy.html
MLA (Modern Language Association) "Enthalpy Change." ScienceAid, scienceaid.net/chemistry/physical/enthalpy.html Accessed 28 Apr 2024.
Chicago / Turabian ScienceAid.net. "Enthalpy Change." Accessed Apr 28, 2024. https://scienceaid.net/chemistry/physical/enthalpy.html.
If you have problems with any of the steps in this article, please ask a question for more help, or post in the comments section below.
Comments
Article Info
Categories : Physical
Recent edits by: Jamie (ScienceAid Editor)