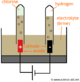
Blast Furnace
Edited by Jamie (ScienceAid Editor), Taylor (ScienceAid Editor), vcdanht, Doug Collins and 6 others
Blasts furnaces are used for smelting during the production of metals such as iron, lead, zinc, and copper. A blast furnace works by blasting hot air up through the furnace while fuels, limestone, and ore move continuously through the top of the furnace. The method used in blast furnaces have been used since the 1500's. Today there are three kinds of blast furnaces used to create metals:
- 1
- 2
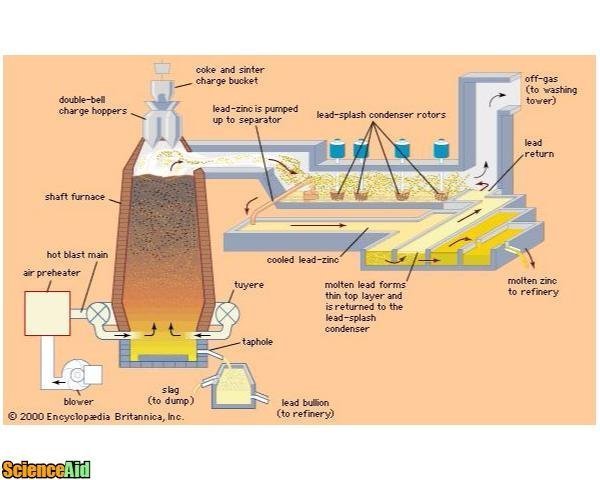
- 3These furnaces are fully sealed and have higher blast rates than lead furnaces.Zinc Blast Furnaces.
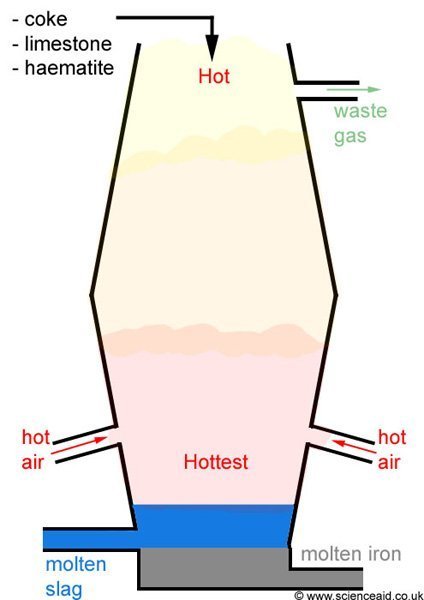
Iron Blast Furnace
What's Happening
In the blast furnace, there are several chemical reactions taking place; that eventually result in the desired product (iron) being extracted. The coke (carbon) burns with oxygen to produce carbon dioxide. This reaction is exothermic. The CO2 then reacts with more coke to give carbon monoxide.
Carbon monoxide acts as a reducing agent and reacts with the iron ore to give molten iron, which trickles to the bottom of the furnace where it is collected.
The limestone in the furnace decomposes, forming calcium oxide. This is a fluxing agent and combines with impurities to make slag, which floats on top of the molten iron and can be removed.
Questions and Answers
Can you give me easy facts about blast furnace or some interesting facts?
We are doing a presentation on blast furnace and we need your help, Would you mind helping us with a presentation? We don't know anything about blast furnaces.
To prepare for your presentation first make an outline. In your outline you can put:
- Why blast furnace is used?
- How iron is formed in the blast furnace?
- Benefits of using blast furnace.
Then,
- In the introduction part, talk about the role of a blast furnace in the formation and collection of iron, and put some pictures about iron and the blast furnace.
- In the body part, talk about the process of formation of iron. Explain what happens inside the blast furnace step by step and put images illustrating the process.
- In the conclusion, talk about the importance of this method and how it is very efficient.
Referencing this Article
If you need to reference this article in your work, you can copy-paste the following depending on your required format:
APA (American Psychological Association)
Blast Furnace. (2017). In ScienceAid. Retrieved Apr 26, 2024, from https://scienceaid.net/chemistry/applied/blastfurnace.html
MLA (Modern Language Association) "Blast Furnace." ScienceAid, scienceaid.net/chemistry/applied/blastfurnace.html Accessed 26 Apr 2024.
Chicago / Turabian ScienceAid.net. "Blast Furnace." Accessed Apr 26, 2024. https://scienceaid.net/chemistry/applied/blastfurnace.html.
If you have problems with any of the steps in this article, please ask a question for more help, or post in the comments section below.
Comments
Article Info
Categories : Applied
Recent edits by: SarMal, Jen Moreau, big boy joshie